The Latest Results from Research & Development
For over 20 years biolitec® has been working closely with leading physicians and medical institutions all over the world to improve minimally invasive laser therapies.
Physicians, who have been using the biolitec® laser therapies for years, are an important part in the company's product developments. A lot of physicians are conducting medical studies and trials in which long-term effects of the biolitec® therapies are analyzed.
Now we would like to present to you the latest results and invite you to contact us to share your experiences and discuss them with us.
Special Focus:
Acne Inversa / Hidradenitis Suppurativa
February 2023:
Guillem, P. et al: "Ultrasonography-guided intralesional diode laser for the treatment of Hurley II hidradenitis suppurativa: Results from a pilot study with 46 procedures", Abstract on 12th Conference of the EHSF - European Hidradenitis Suppurativa Foundation e.V. on 8th Febr. - 10th Febr. 2023 in Florence, Italy (PDF here)
February 2023:
Medhat, M.M. et al: "Hidradenitis suppurativa: interstitial laser treatment with Endolift® procedure using Eufoton® LASEmaR®1500 1470-nm wavelength", in: Journal of Applied Cosmetology Vol. 41 No 1 (2023). https://doi.org/10.56609/jac.v41i1.68
July 2020:
Karabay, Ö.: „First Clinical Experience of FiLaC TM in Hidradenitis Suppurativa: Is it a Safe and Feasible Treatment Modality?“, in: Turkish Journal of Colorectal Disease 2020;30(4):296-300. https://doi.org/10.4274/tjcd.galenos.2020.2020-6-7
Special Focus:
FiLaC® and Morbus Crohn
The minimally invasive, sphincter-sparing laser therapy FiLaC® (Fistula-tract Laser Closure) enables excellent healing results in the treatment of complex anal fistulas with maximum sparing of the surrounding tissue and is thus comparable to other sphincter-sparing techniques. This technique is therefore excellently designed to ensure healing of anal fistulas even in patients with Crohn's disease. This makes it possible for patients, under certain circumstances, to dispense with a long-lasting suture appliance, which they might accept for fear of damaging the sphincter muscle.
In their meta-study, Cao et al identified 6 studies with 50 pfCD patients, and 31 patients’ fistulas were healed after FiLaC®. This is a very high success rate of 68% without negatively affecting continence in any way.
The high rate of Crohn's disease patients and the fact that 43% of these patients will develop an anal fistula one day can be found in the meta-analysis by Ng et al. These data selected in countries worldwide highlight the need for research into prevention of inflammatory bowel disease and innovations in health-care systems to manage this complex and costly disease.
August 2022:
Cao, D. et al: "Efficacy and safety of FiLaC™ for perianal fistulizing Crohn’s disease: a systematic review and meta-analysis", in: Techniques in Coloproctology 2022, 26, pages 775–781. https://doi.org/10.1007/s10151-022-02682-1
April 2021:
Nordholm-Carstensen, Andreas et al: „Fistula Laser Closure (FiLaC™) for fistula-in-ano—yet another technique with 50% healing rates?“, in: International Journal of Colorectal Disease 2021, 36, pages 1831–1837. https://doi.org/10.1007/s00384-021-03932-8
Published December 2019:
Alam, A., Lin, F., Fathallah, N. et al: „FiLaC® and Crohn’s disease perianal fistulas: a pilot study of 20 consecutive patients“, in: Techniques in Coloproctology 2020, 24, pages 75–78. https://doi.org/10.1007/s10151-019-02134-3
October 2017:
Ng, Siew C. et al: "Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies", in The Lancet 2017, volume 390, issue 10114, pages 2769–2778. https://doi.org/10.1016/S0140-6736(17)32448-0

LHP® for Hemorrhoids
In addition to several case series, the Laser Hemorrhoidoplasty (LHP®) was also the subject of comparative studies. Poskus and Eskandaros et al have been able to demonstrate in their studies that the laser is extremely painless and causes fewer complications than the hemorrhoidopexy, Milligan Morgan or HAL/RAR in the treatment of hemorrhoidal disorders. Also, the patient’s outcome in terms of quality of life was in favor of the LHP® procedure.
April 2023:
Gambardella, C., Brusciano, L., Brillantino, A. et al.: "Mid-term efficacy and postoperative wound management of laser hemorrhoidoplasty (LHP) vs conventional excisional hemorrhoidectomy in grade III hemorrhoidal disease: the twisting trend", in: Langenbeck's Archive of Surgery 408, 140 (2023). https://doi.org/10.1007/s00423-023-02879-4
July 2021:
Majumder, K. R. et al: "LASER Haemorrhoidoplasty versus Stapler Haemorrhoidopexy: A Prospective Comparative Study", in: Mymensingh Medical Journal 2021,30(3):780-788. https://pubmed.ncbi.nlm.nih.gov/34226468/
April 2020:
Eskandaros, Moheb S. and Darwish, Ahmed A.: "Comparative study between Milligan-Morgan hemorrhoidectomy, stapled hemorrhoidopexy and laser hemorrhoidoplasty in patients with third degree hemorrhoids: a prospective study", in: The Egyptian Journal of Surgery 2020, 39:352-363. https://doi.org/10.4103/ejs.ejs_214_19
January 2020:
Poskus, Tomas et al: "Results of the double-blind randomized controlled trial comparing laser hemorrhoidoplasty with sutured mucopexy and excisional hemorrhoidectomy", in: International Journal of Colorectal Disease 2020, 35(3):481-490. https://doi.org/10.1007/s00384-019-03460-6
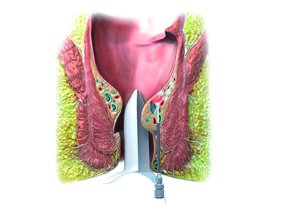
FiLaC® for Anal Fistulas
Professor Giamundo recently presented his ten-year data for anal fistulas therapy. At a median follow-up of five years, his group had a success rate of 67%, paired with a very low complication rate. The meta-analyses by Elfeki and Frountzas et al came to similar results with nearly 1000 patients included. The great advantage is that the FiLaC® procedure does not change the continence adversely. Furthermore, it has been successfully demonstrated that both, complex fistulas that have been operated several times before and Crohn's disease fistulas (Alam et al), can successfully be treated with this laser procedure. With regard to the individual approach, this procedure offers a very high degree of freedom with a wide range of fistula applications at the same time.
May 2021:
Giamundo, Paolo and Angelis, Marsilio De: "Treatment of anal fistula with FiLaC®: results of a 10-year experience with 175 patients", in: Techniques in Coloproctology 2021, 25, pages 941–948. https://doi.org/10.1007/s10151-021-02461-4
December 2020:
Frountzas, M. et al: "Could FiLaC™ be effective in the treatment of anal fistulas? A systematic review of observational studies and proportional meta-analysis", in: Colorectal Disease 2020, 22(12):1874-1884. https://doi.org/10.1111/codi.15148
April 2020:
Elfeki, H. et al: "A systematic review and meta-analysis of the safety and efficacy of fistula laser closure", in: Techniques in Coloproctology 2020, 24(4):265-274. https://doi.org/10.1007/s10151-020-02165-1
March 2017:
Wilhelm, A. et al: "Five years of experience with the FiLaCTM laser for fistula-in-ano management: long-term follow-up from a single institution", in: Techniques in Coloproctology 2017, 21:269–276. https://doi.org/10.1007/s10151-017-1599-7

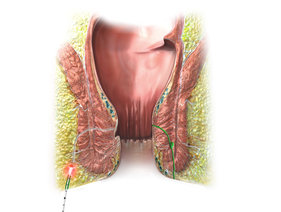
SiLaC® for Sinus Pilonidalis
Based on a large number of publications on the topic of coccyx treatment with the laser, it can be stated that patients can be treated within a few minutes with a very high success rate of more than 90%. The systematic review by Romic et al proves this in an overview paper. In contrast, open excision has a very high rate of wound healing disorders and a long convalescence. Skin flap plastics on the other hand often lead to an unwanted cosmetic result. With the SiLaC® procedure, all inflammatory fistula tracts can be destroyed through the smallest of accesses and hair and granulation tissue can be removed for cleaning. The rima ani is spared as much as possible and only very small wounds (less than 4 millimetres) are created.
March 2023:
Li, Z., Jin, L., Gong, T. et al.: "An effective and considerable treatment of pilonidal sinus disease by laser ablation", Lasers in Medical Science 2023, 38, Article number 82 (2023). https://doi.org/10.1007/s10103-023-03741-1
August 2021:
Petersen, Sven et al: "[Wound Healing Disorders after Excision and Open Treatment for Pilonidal Sinus]" [article in German], in: Zentralblatt für Chirurgie 2021, 146(04): 417-426. https://doi.org/10.1055/a-1301-2051
July 2021:
Romic, Ivan et al: "Laser treatment of pilonidal disease: a systematic review", in: Lasers in Medical Science (2021). https://doi.org/10.1007/s10103-021-03379-x
January 2021:
Danys, Donatas et al: "SMILE technique for Pilonidal Sinus destruction with a radial laser probe - video vignette", in: Colorectal Disease 2021, 23(4):1023-1024. https://doi.org/10.1111/codi.15518
Contact
biolitec®
Otto-Schott-Str. 15
07745 Jena
Germany
Tel:
+49 (0) 3641 519 53 0
Fax:
+49 (0) 3641 519 53 33
E-Mail:
info[at]biolitec.de
Internet:
www.biolitec.com